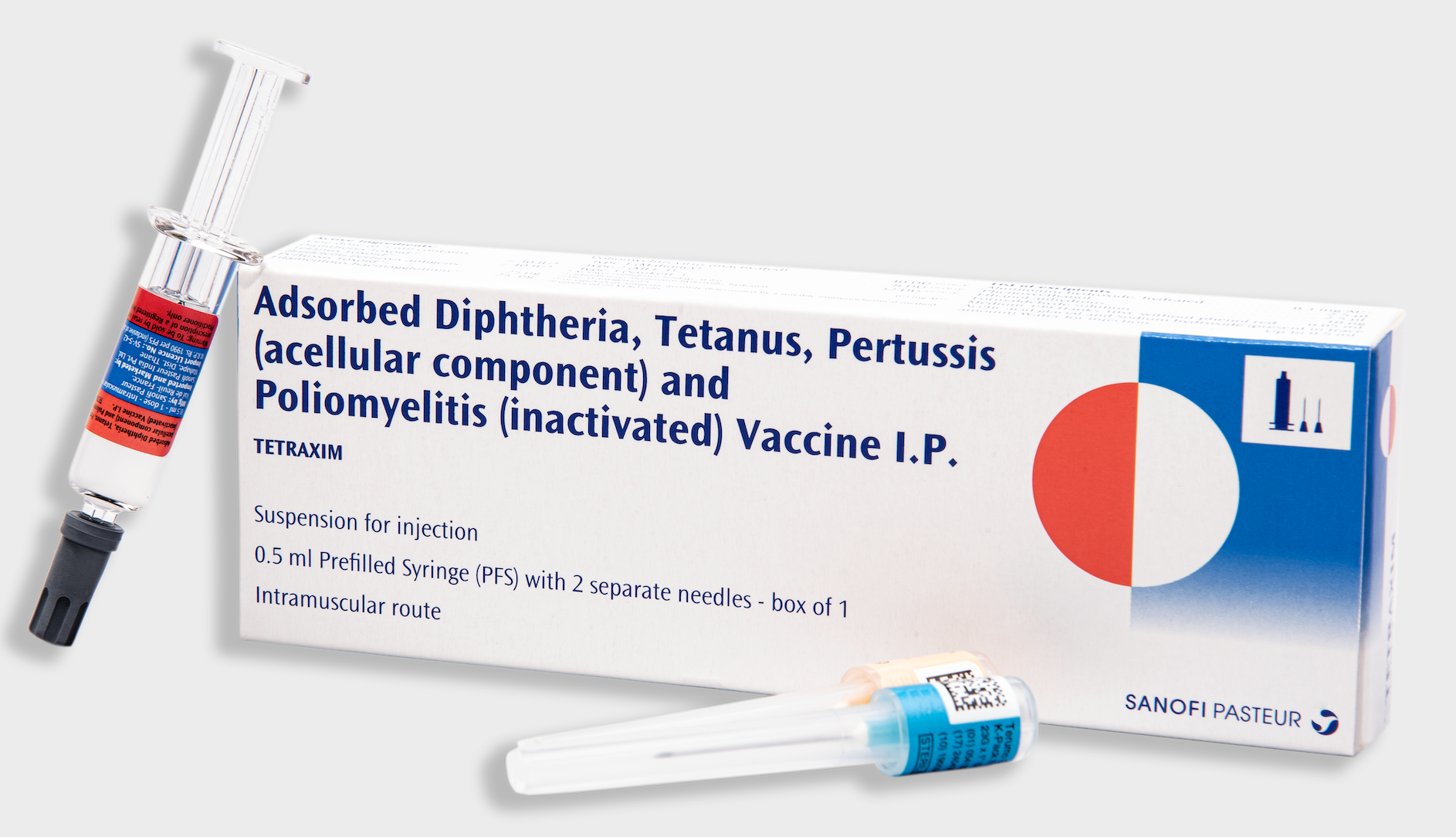
Sanofi Pasteur launches Tetraxim, the first full-dose DTaP booster vaccine in India for preschoolers, ET HealthWorld

Safety and immunogenicity of inactivated poliovirus vaccine schedules for the post-eradication era: a randomised open-label, multicentre, phase 3, non-inferiority trial - The Lancet Infectious Diseases
![PDF] Poliomyelitis prevention in the United States: introduction of a sequential vaccination schedule of inactivated poliovirus vaccine followed by oral poliovirus vaccine. Recommendations of the Advisory Committee on Immunization Practices (ACIP) PDF] Poliomyelitis prevention in the United States: introduction of a sequential vaccination schedule of inactivated poliovirus vaccine followed by oral poliovirus vaccine. Recommendations of the Advisory Committee on Immunization Practices (ACIP)](https://d3i71xaburhd42.cloudfront.net/5c888623f677b8556e9175d52fb34446f36edd7a/20-Table4-1.png)
PDF] Poliomyelitis prevention in the United States: introduction of a sequential vaccination schedule of inactivated poliovirus vaccine followed by oral poliovirus vaccine. Recommendations of the Advisory Committee on Immunization Practices (ACIP)
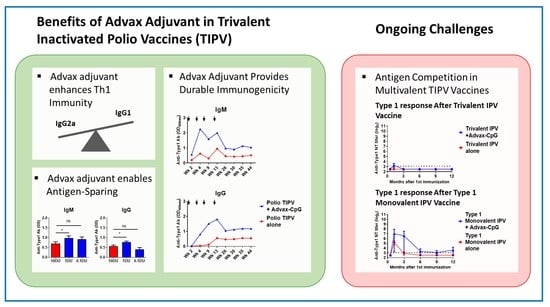

![PDF] Fractional doses of inactivated poliovirus vaccine in Oman. | Semantic Scholar PDF] Fractional doses of inactivated poliovirus vaccine in Oman. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0d86345fa31e73d91db5772400d41eafea9eee02/5-Table1-1.png)

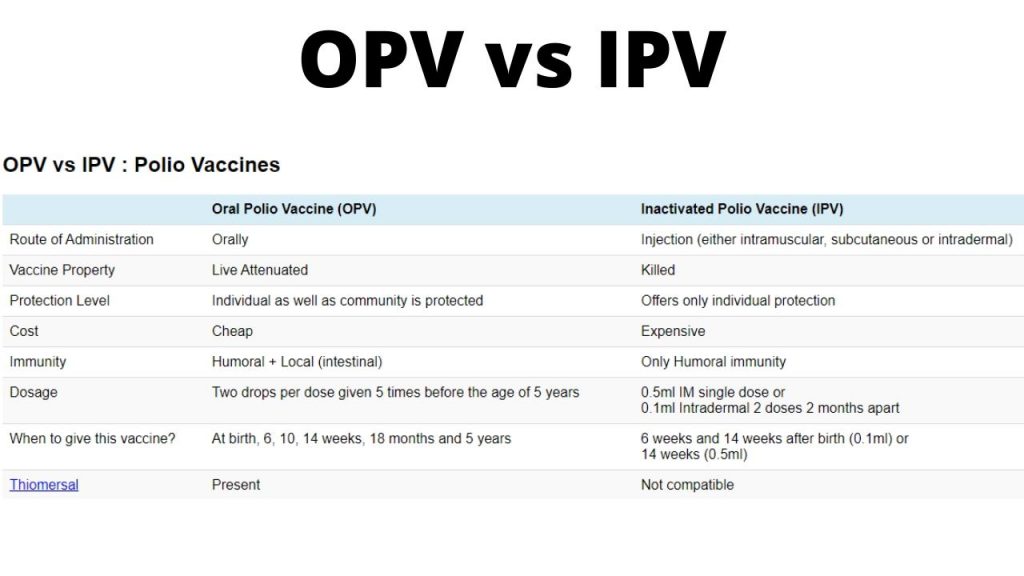



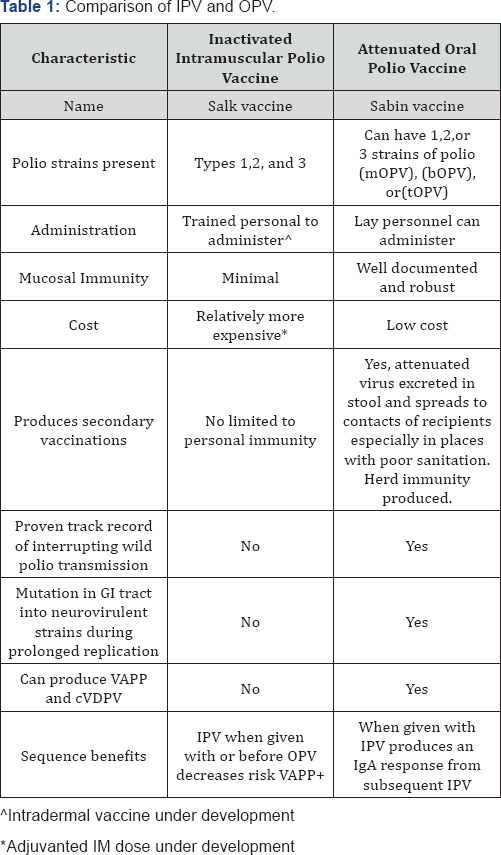


![PDF] Polio vaccination: past, present and future. | Semantic Scholar PDF] Polio vaccination: past, present and future. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/998a3b03dff345207ff5176c107f25f3e0c773c7/9-Table1-1.png)